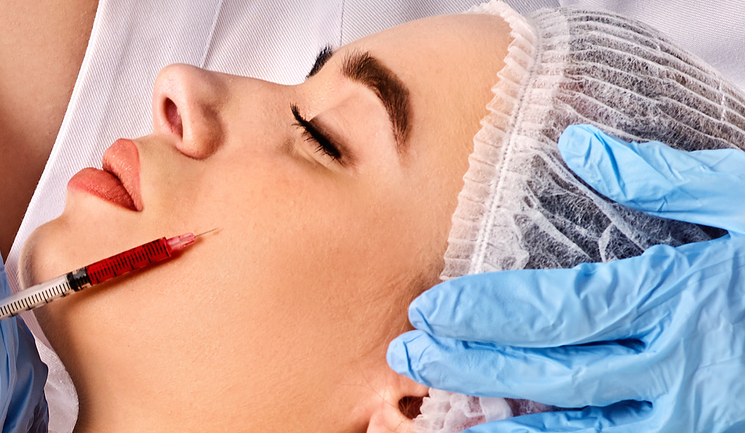
Dr Zunaid Alli shares a filler complication case study and his steps for management
To access this post, you must purchase Aesthetics Journal Membership – Annual Elite Membership, Aesthetics Journal Membership – Annual Enhanced Membership or Aesthetics Journal Membership – Basic Membership.
log in
log in