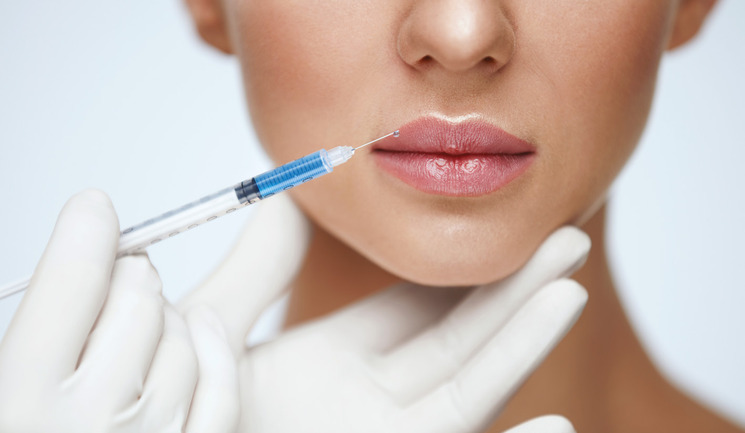
Dr Saleena Zimri discusses the treatment of patients with hyaluronic acid after previous dissolution and shares three successful case studies
To access this post, you must purchase Aesthetics Journal Membership – Annual Elite Membership, Aesthetics Journal Membership – Annual Enhanced Membership or Aesthetics Journal Membership – Basic Membership.
log in
log in