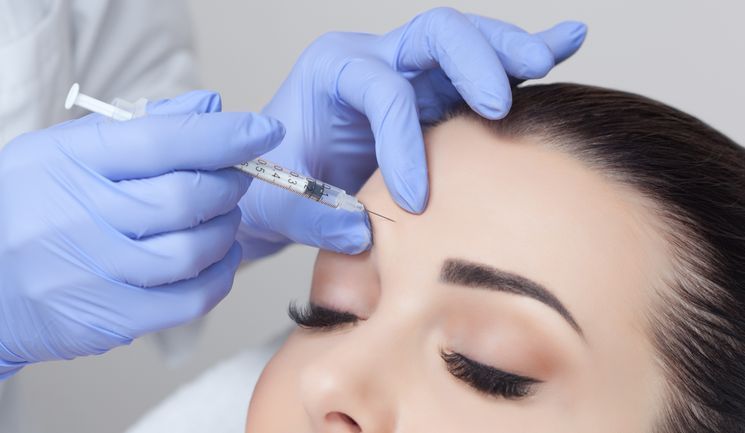
Global pharmaceutical company Galderma has received top-line results for its phase 2 study in the US on the efficacy and safety of a single dose of abobotulinumtoxinA versus placebo for the treatment of moderate to severe glabellar lines.
Researches treated 401 subjects aged 18 to 65 with moderate to severe glabellar lines at maximum frown using Dysport – also known as Azzalure in the UK. Study participants were randomised 4:1 to receive a single dose of Dysport or placebo and followed for nine months.
Results showed that the study met its primary endpoint, with significantly more subjects treated with Dysport at all four doses achieving a composite two-grade improvement responder rate than those treated with placebo at one month.
Dr John Joseph, investigator for the study and director of The Clinical Testing Center of Beverly Hills said, “We are encouraged that these study results demonstrate that a single dose of Dysport has a rapid onset, long-lasting effect and is well tolerated. At all doses tested in the trial, Dysport had a strong safety profile, including very few cases of eyelid ptosis.”