
FDA warns against needle-free injector pens
The US Food and Drug Administration (FDA) has issued a safety communication regarding the use of needle-free injection devices for fillers.

The US Food and Drug Administration (FDA) has issued a safety communication regarding the use of needle-free injection devices for fillers.

Dr Dev Patel discusses treating young patients and why he believes the term ‘filler’ is no longer relevant, especially when looking to achieve natural looking results for this patient demographic
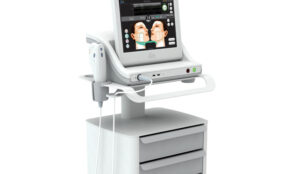
Aesthetic nurse Anna Gunning explains how you can be one step ahead of the game and invest in devices for optimum results

Medical device manufacturer Prollenium has launched its new dermal filler brand Revanesse in the UK.

Biopharmaceutical company Hugel America and aesthetic manufacturer Croma-Pharma has unveiled that the US Food and Drug Administration (FDA) has approved the Biologics License Application (BLA) for letibotulinumtoxinA as a treatment for glabellar lines.

UK manufacturer Laser Lipo has expanded the distribution of its Strawberry Laser to the Scandinavian region by partnering with health and wellbeing company Top Motion.
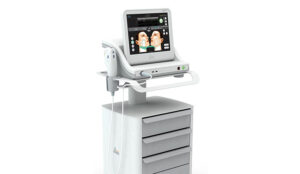
How technology that lets you see under the skin can optimise lifting and tightening results

Dr Selena Langdon shares her most popular pre-summer body treatment tips

Aesthetic practitioner and owner of PHI Clinic in London Dr Tapan Patel is a new UK key opinion leader for skin tightening laser treatment Endolift.

Skin device Tixel, manufactured by Novoxel, has received clearance from the Food and Drug Administration (FDA) for dermatological procedures requiring ablation and resurfacing of the skin.