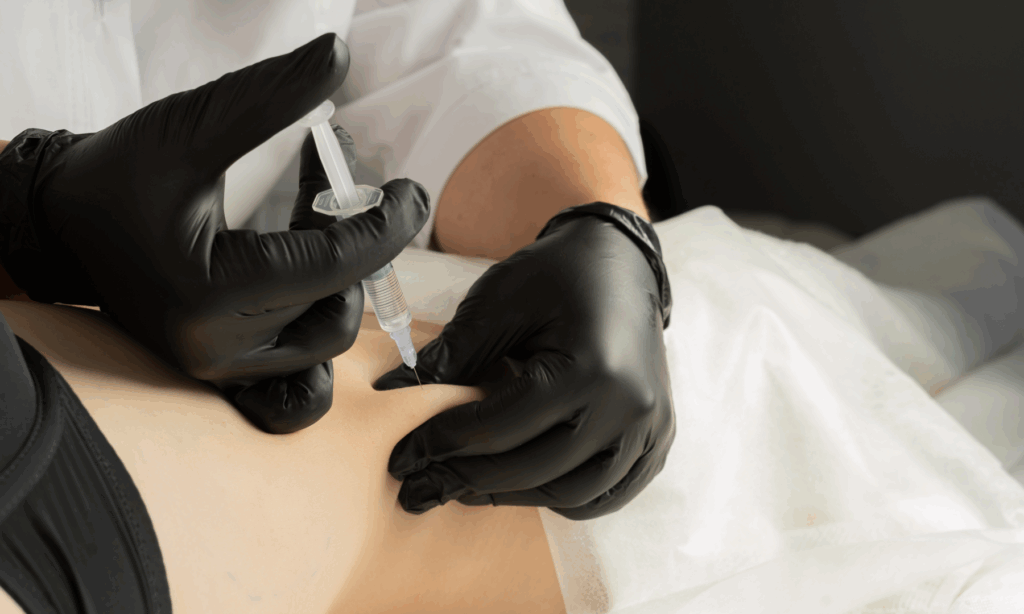
The Joint Council for Cosmetic Practitioners (JCCP) has issued an advisory warning against the use of phosphatidyl choline (PPC)-based products in cosmetic practice.
The organisation explains that the use of PPC-based products is limited to the treatment of medically diagnosed indications, such as the treatment of lipoma.
The JCCP shares that PCC is not authorised for cosmetic use in the UK, according to confirmation from the Medicines and Healthcare products Regulatory Agency (MHRA).
This advisory therefore applies to all individuals who use or supply these products, as well as those responsible for regulating or investigating practitioners or organisations, including local authority officers. As a result, the JCCP has requested all JCCP registrants to cease using these products for cosmetic purposes or promoting their use in any circumstances.
A spokesperson from the JCCP commented, “This advisory makes it clear that, to comply with medicines legislation, practitioners must not use these PPC based products for cosmetic purposes. Evidence across the cosmetic sector increasingly points to the risks to public safety, and to professional reputation, when products are sourced and administered inappropriately. We hope that this advice serves as a reminder to all practitioners to consider carefully the products they use and how they obtain them. We also expect it to assist enforcement officers in there duties, and to inform the ongoing development of a framework of licensing.”